Spotlight on the physician perspective: Conversation with Dr. Ariel Pablos-Méndez
On Monday, March 30, Rabin Martin, as the Secretariat for the Private Sector Roundtable for Global Health Security (PSRT), hosted a sixth call in a series on the latest health impacts of COVID-19. Dr. Ariel Pablos-Méndez has had a distinguished career in global public health, with roles at the World Health Organization, Rockfeller Foundation, USAID and Columbia University Irving Medical Center, where he is now professor of medicine and internist. Dr. Pablos- Méndez provided his insights on the current situation in New York City – the new global epicenter. Dr. Pablos-Méndez emphasized that this is a hyperacute epidemic and that we’re only seeing the “tip of the iceberg.” He expects ICUs in NYC to be overwhelmed for another couple of months.
Notably, he urged greater attention to testing to understand fully the extent of the epidemic and its trajectory – and argued that serology is more important now than vaccines because, in most places, the virus will be gone before a vaccine is developed. Testing is also important because he estimates that a high proportion of health care workers may already be positive and, if they were tested, they would be relieved to know they were immune and could continue working. Finally, Dr. Pablos-Méndez recommended an age-bifurcated approach to the response, calling for “obsessively isolating the elderly and the sick” given that 90% of deaths have been among those over 65 and those who have other illnesses. He added that the situation is quite different from the H1N1 virus, for example, which led to the deaths of many children.
Spotlight on the patient perspective: Q&A with The Mighty
Rabin Martin spoke with Mike Porath, Founder and CEO of The Mighty – an online patient community of more than two million registered users facing health challenges around the world. On March 26, The Mighty published findings from a survey on the healthcare impact of COVID-19 amongst its users. 72 percent of respondents reported that their healthcare has changed because of the on-going crisis; 35 percent have gone without doctor visits; and 23 percent have gone without medications or treatments. Our discussion focused on exploring these findings and how decision makers can help mitigate the crisis.
What are the most surprising trends you’re seeing among patients and their reaction to COVID-19?
Fear and anxiety over this pandemic have really skyrocketed over the last couple weeks, though that’s not surprising. What’s most striking to me is the impact to people’s healthcare. More than a third of our community members are reporting they aren’t able to see their doctors. Nearly a quarter aren’t getting their medications or treatments.
How do you think these patients’ views compare with those of the overall population given general heightened anxiety?
Our community is made up of patients, caregivers and healthcare providers. We have a lot of high-risk people who are taking more actionable steps to avoid contracting the virus than the average population. They are also more concerned about the long-term effects of this outbreak – the inability to get the healthcare they need. I’m not surprised that their anxiety levels are through the roof right now.
What differences are you seeing based on disease or condition? Are patients with certain conditions responding differently than others?
The clearest difference is that people who are at high risk for contracting coronavirus – people who are immuno-compromised – are more anxious and fearful and have really changed their behaviors to limit their exposure. For people managing mental health issues, this is a very difficult time. It can be paralyzing.
Given the findings, what would you like to see from health care leaders – government, health care companies, health care providers, hospitals – do to support patients during this crisis?
I would like to see the focus return to the patient. Let’s put the “care” back in healthcare. This pandemic is causing people to lose access to their healthcare. We asked them what they needed right now. 38% said telehealth services. 31% said a delivery service from pharmacies. If every organization in healthcare focused on delivering for patients, we’d do a better job of helping them. And that’s what healthcare is all about.
Pandemic Spread
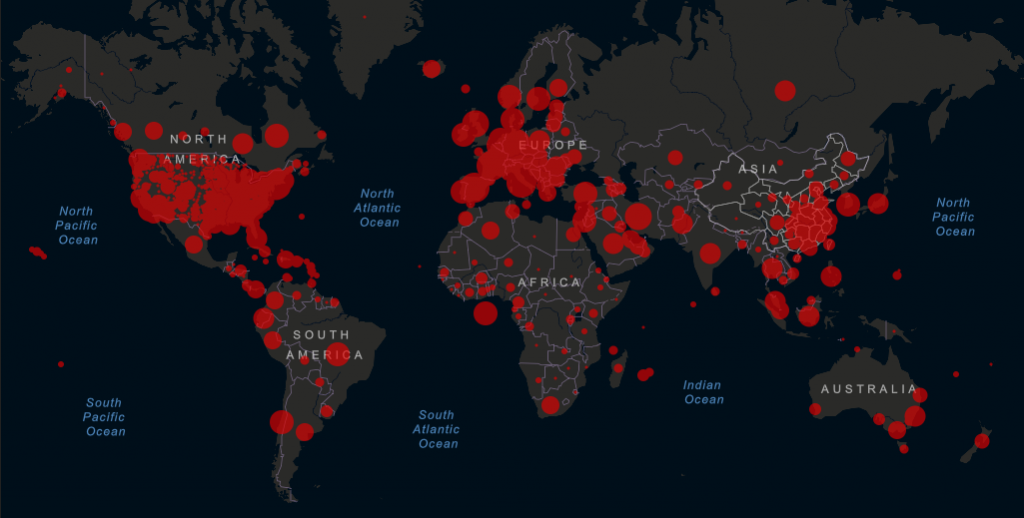
In the past week, the virus has spread exponentially throughout the U.S. to become the largest epidemic of any country in the world. There are now 217,263 reported cases of COVID-19 in the U.S., twice as many cases as seen in Italy, the second-hardest hit country. U.S. cases account for nearly a quarter of the global burden.
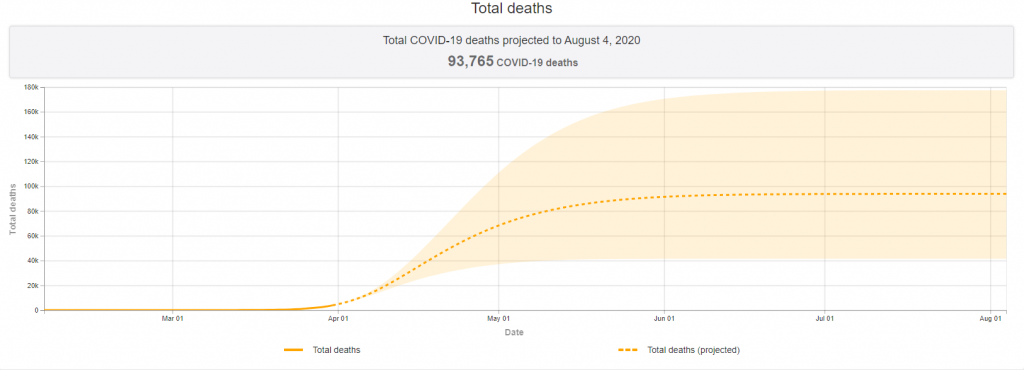
Models by the Institute for Health Metrics and Evaluation suggest current diagnoses represent just a fraction of eventual U.S cases. IHME predicts the epidemic will peak in just 13 days, on Wednesday, April 15. At that point, demand for health care resources will far outstrip supply: there could be a shortage of almost 88,000 hospital beds and 32,000 ventilators. Barring new social-distancing interventions or increased resourcing, these projections suggest a poor prognosis for the U.S. The country could see nearly 94,000 deaths in the epidemic’s first wave. Worldwide, as of Thursday, April 2 at 11:00am ET, the Center for Systems Science and Engineering at Johns Hopkins University reported 965,246 confirmed cases and 49,236 deaths attributed to COVID-19.
Notable Industry Developments
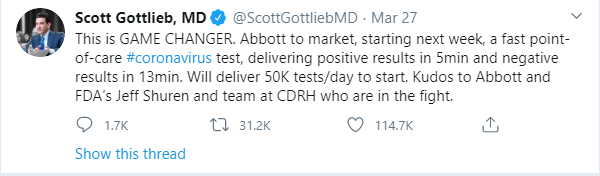
On Friday, March 27, the FDA granted emergency use authorization for Abbott’s COVID-19 test – the fastest available molecular point-of-care test for COVID-19. The test can deliver positive results in as little as five minutes.
On Saturday, March 28, Gilead announced that it was increasing access to its investigational medicine remdesivir (currently in clinical trials) by transitioning from a “compassionate use” program for relatively few patients suffering from COVID-19 to an “expanded access” program, whereby hospitals and physicians can apply for emergency use of the treatment for multiple severely ill patients at a time.
On Monday, March 30, Johnson & Johnson announced that it had selected a lead COVID-19 vaccine candidate and expects to initiate human clinical studies by September 2020. The company also announced an expanded collaboration with the Biomedical Advanced Research and Development Authority (BARDA – a $1 billion commitment to vaccine research, development and clinical testing – and plans to supply one billion vaccines worldwide, beginning with an emergency use authorization anticipated in early 2021.
On Monday, March 30, Novartis announced it would donate 20,000 doses of hydroxychloroquine to the University of Washington for a COVID-19 post-exposure prophylaxis clinical trial. Sandoz, the company’s generics and biosimilars division, is facilitating the donation, with funding from the Bill & Melinda Gates Foundation.
Recent Philanthropic Commitments
The clinical trials required to prove the safety and efficacy of vaccine and treatment candidates are extensive, and the time to bring these products to market is long. Beyond investments in research and development, pharmaceutical companies are committing extensive resources – both monetary and in-kind – to combat the pandemic, supporting the United Nations’ call for an “immediate, coordinated health response.” A comprehensive list may be found here. Select commitments from the past week include:
- On Wednesday, March 25, GSK announced a $10 million donation to The COVID-19 Solidarity Response Fund, created by the UN Foundation and WHO, focusing on distributing essential equipment to healthcare workers.
- On Wednesday, March 25, Takeda announced a $6.25 million donation to organizations in the United States, including the American Red Cross to support the organization in its delivery of essential services nationwide, such as maintaining a sufficient supply of blood for patients in need.
- On Friday, March 27, Bloomberg Philanthropies and the State of Maryland committed $4 million in funding to Johns Hopkins University researchers to explore therapeutic uses of blood plasma for potential treatment for COVID-19.
- On Friday, March 27, Johnson & Johnson announced a $50 million donation to support the needs of frontline healthcare workers, focusing on doctors, nurses, midwives and community health workers.
- On Tuesday, March 31, AbbVie announced a $35 million donation to Direct Relief, Feeding America and International Medical Corps. AbbVie’s funds will be used to support healthcare capacity for hospitals, enable access to food and provide equipment and supplies to patients and healthcare workers.
- On Tuesday, March 31, Amgen announced a donation of up to $12.5 million to support emergency response efforts by patient-focused organizations and international relief efforts by Direct Relief and International Medical Corps.
From the Experts
“Previous outbreaks have demonstrated that when health systems are overwhelmed,deaths due to vaccine-preventable and treatable conditions increase dramatically. Even though we’re in the midst of a crisis, essential health services must continue. Babies are still being born, vaccines must still be delivered, and people still need life-saving treatment for a range of other diseases.”
– Dr. Tedros Adhanom Ghebreyesus, WHO Director-General
Monday, March 30
“What we’re going through now is going to be more than just lessons learned; it’s going to be things that we have available to us that we did not have before.”
– Dr. Anthony Fauci, Director
National Institute of Allergy and Infectious Disease
Monday, March 30
“We must respond decisively, innovatively and together to suppress the spread of the virus and address the socio-economic devastation that COVID-19 is causing in all regions.“
– Antonio Guterres, Secretary-General, United Nations
Tuesday, March 31
“We remain very concerned about the negative outlook for global growth in 2020 and in particular about the strain a downturn would have on emerging markets and low-income countries. Our forecast of a recovery next year hinges on how we manage to contain the virus and reduce the level of uncertainty.”
– Kristalina Georgieva, Managing Director
International Monetary Fund
“There’s no magic bullet. There’s no magic vaccine or therapy. It’s just behaviors. Each of our behaviors translating into something that changes the course of this viral pandemic over the next 30 days.”
– Ambassador Deborah Birx, Coordinator, United States Government Activities to Combat
HIV/AIDS, and White House Coronavirus Response Coordinator
Tuesday, March 31
In Memoriam: Professor Gita Ramjee

The COVID-19 pandemic does not discriminate. On Wednesday, April 1, it took one of the global health community’s most admired researchers and advocates: Professor Gita Ramjee, a leading South African scientist. Gita dedicated her career to the fight against HIV/AIDS, especially new methods of prevention for women. All of us at Rabin Martin are grateful for her contributions to end the HIV/AIDS epidemic, and will continue to be inspired by her intellect and passion.
Additional Resources
Reports from International Governments and Bodies
- WHO COVID-19 Information and Guidance
- WHO Situation Reports, March 30, March 31, April 1
- White House Coronavirus Task Force Press Briefings, March 30, March 31
- CDC Coronavirus Resource Page
- NCD Alliance COVID resources relevant to NCDs
Funding and Policy Trackers
- International Monetary Fund Policy Tracker
- Kaiser Family Foundation Coronavirus Policy Tracker
- U.S. Chamber of Commerce Foundation Corporate Aid Tracker
- Devex Interactive Funding Tracker
- Page Society COVID-19 Toolkit
Academic and Market Research Literature
What We’re Reading
COVID-19: Now is the time to create the future we want, Mark Dybul and Deus Bazira, The Hill
Can Deborah Birx save us? Emily Bass, Washington Post
It’s a razor’s edge we’re walking: inside the race to develop a coronavirus vaccine, Samanth Subramanian, The Guardian
Developing Covid-19 vaccines at pandemic speed, Nicole Lurie, Melanie Saville, Richard Hatchett, Jane Halton, NEJM
“Take as Directed: Coronavirus Crisis Update” Podcast, CSIS
Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma, Chenguang Shen, Zhaoqin Wang, Fang Zhao, NEJM